Ciclosporina, Antralina, Minoxidil y antralina, ¿Es útil tratar a los pacientes que no responden?, Tratamiento de los niños con difenciprona. Guías terapéuticas de la AAD para la Alopecia areata. Tratamiento con inmunoterapia: Revisión de 129 pacientes, Alopecia areata provocada por vacunación, Zinc en la Alopecia areata, Difenciprona: experiencia con 75 pacientes, Discromia en confeti con difenciprona, Rol de la inmunoterapia en la AA: revisión de los trabajos publicados entre 1977 y 1988, Tratamiento de la alopecia areata de larga duración, FK506 tópico, New treatments for alopecia areata, Protocolo de tratamiento de la alopecia areata de la UCSF-UCB , Topical immunotherapy of severe alopecia areata with diphenylcyclopropenone: Evaluation of 68 cases Pulse methylprednisolone therapy for severe alopecia areata: an open prospective study of 45 patients PUVA Treatment for Alopecia areata Dermatology 197:3:1998, 245-247 Price VH. Treatment of hair loss. NEJM 1999 341:964, Predictive Model for Immunotherapy of Alopecia Areata With Diphencyprone The use of methotrexate
Gupta AK. Oral cyclosporine for the treatment of alopecia areata. JAAD 1990 22:242-50
we treated six patients with oral cyclosporine 6 mgr/kg/dia/`12w. three had alopecia universalis one had alopecia totalis and two had patchy alopecia areata of the scalp. Hair re growth in the scalp occurred within the second and four weeks of therapy, followed by hair re growth of the face and chest(on male patients), pubic area, extremities, and axillae, overall the best response was the scalp. Cosmetically acceptable hair re growth occurred in three of the six patients.Baseline
|
perdida de pelo |
semana 12 | semana 14 |
| 25-74% | 6/16(38%) | 10/15(67%) |
| 75-99 | 3/19(16%) | 5/16(31%) |
| 100% | 1/15 | 2/14(14%) |
Treatment of resistant alopecia areata. Fridler VC et al. Arch Dermatol
1990, 126:756-9.
Minoxidil al 5%, después de la aplicación
de 1 mil de Minoxidil cada 12 horas, 2 horas después de la aplicación
del Minoxidil aplicar 1 gr. de antralina al 0.5% que se debe dejar toda
la noche, respuesta moderada a cosmética en la combinación
de Minoxidil y antralina
Fiedler Weiss VC. Evaluation of antralin
in the treatment of alopecia areata. AD 1987 123:1491-3
Las aplicación fueron hechas al acostarse a días
alternos durante las dos primeras semanas, entonces diariamente, a las
8 horas se lavaban la cabeza con un champú de ptitiriona de zn.
La duración media del tratamiento fue de 28 semanas (8-200)
|
|
||||
|
|
|
|
||
|
|
|
Pt | respuesta cosmetica | |
| 0-24%
25-75% 75100% |
4
33 29 |
1(25%)
8(25%) 4(14%) |
4
34 30 |
1(25%)
10(29%) 6(20%) |
Tosti A. et al. Alopecia totalis: is treating nonresponder
patients useful?. JAAD 1991 24:455-6.
26 patients who had been receiving sensitizing therapy
for at least 1 year without any response were randomly assigned to receive
the following treatments:
Macdonald S, Pepall L, Cunliffe WJ. Alopecia areata in children: response to tratment with dephencyprone. Br J Dermatol 1991, 125: 164-8.
Doce niños con alopecia areata extensa o alopecia total fueron tratados con el alergeno de contacto difenciprona. La duración del tratamiento duró entre 5 meses y un año. 8 de los 12 mostraron crecimiento del cabello y en 4 (33%) el crecimiento fue completo. 6 meses después de cesar el tratamiento 3 de los 4 niños con crecimiento completo del cabello mantenían su cabello y uno había perdido todo el cabello que había ganado, y un chico que había presentado solo un crecimiento parcial presentó un crecimiento total.
Drake L, Ceilley R, Cirnelison R, Dobes W, Dorner W, Goltz R, Lewis C, Salasche S, Chanco ML. Guidelines of care for alopecia areata. J Am Acad Dermatol 1992, 26: 247-250.
Introducción: la AAD va a desarrollar unas guías
de tratamiento que ayuden a dar un tratamiento de calidad y ayuden a otros
especialistas a comprender la complejidad del tratamiento instaurado por
estos.
Definición: La alopecia areata es una enfermedad
que afecta al cabello y en ocasiones a las uñas, Esta es una enfermedad
que puede ocurrir de forma aislada o asociada a otras condiciones médicas
variadas. La etiología de la alopecia areata es desconocida pero
está caracterizada por una disfunción del folículo
piloso y por la presencia de un infiltrado monocuclear peribulbar y perifolicular.
La enfermedad puede afectar a cualquier zona del cuerpo cubierta por cabello
pero es más frecuentemente observada en el cuero cabelludo, párpados
y barba.
Situación actual: La alopecia areata es
una enfermedad frecuente con una prevalencia de uno cada 1000. Sin embargo
la prevalenc exacta es desconocida, principalmente ya que los casos más
moderados nunca llaman la atención. No existe predilección
por la edad, seco o ocupació profesional. La alopecia areata puede
ocurrir a cualquier edad y el curso es altamente variable. Las recidivas
son frecuentes y la mayor parte de los pacientes pueden tener recidivas.
La causa de la alopecia areata es desconocida. Los últimos
datos sugieren que es una enfermedad mediada inmunológicamentes.
En la alopecia areata no existe una destrucción permanente de los
folículos pilosos. En ocasiones hay asociadas otras enfermedades
autoinmunes tales como las tiroiditis erc.
Criterios diagnósticos
1-Clínicos
alopecia areta del cuero cabelludo
Factores de posible significancia pronostica en la immnoterapia topica de la alopecia areata
tipo de afectación % de respuesta
Macdonald S, Cunliffe WJ. Succesful treatmen of alopecia areata using the contact allergen diphencyprone. Br J Med 1991, 124:212-3.
78 adultos 17_ 61_, con AA duración media de 7 años (0.5-35), después de 8 meses de tratamiento:
29 (22AT, 7AA)
sin crecimiento
49 (23AT,
26AA) crecimiento
25/49 crecimiento
completo (18AA, 7AT)
van-der-Steen-P; Happle-R.'Dyschromia in confetti' as a side effect of topical immunotherapy with diphenylcyclopropenone. Arch-Dermatol. 1992 Apr; 128(4): 518-20
"Dyschromia in confetti" is a pigmentary disturbance that so far has not been reported to have occurred during topical immunotherapy. In a group of 243 patients treated with diphenylcyclopropenone for alopecia areata, four patients showed a similar pattern of hyperpigmentation and hypopigmentation. In one patient the pigmentary disturbance was confined to the site of diphenylcyclopropenone application on the scalp. Besides dyschromic skin changes on her scalp, one patient had development of a hyperpigmentation on her forearms. In the remaining two patients, dyschromia in confetti became manifest exclusively in areas other than the scalp. This type of pigmentary disturbance appeared 4 to 14 months after the start of treatment, and it has remained unchanged in three patients during a follow-up period of 3 to 10 months. In one patient, the pigmentary changes disappeared when therapy was discontinued but recurred, albeit to a lesser extent, after treatment had been resumed. We conclude that dyschromia in confetti should be regarded as a possible, but probably rare, side effect of topical immunotherapy with diphenylcyclopropenone, especially in individuals with a rather dark complexion.
Berth-Jones-J; Hutchinson-PE Treatment of alopecia totalis with a combination of inosine pranobex and diphencyprone compared to each treatment alone. Clin-Exp-Dermatol. 1991 May; 16(3): 172-5
Recent developments in alopecia areata have included the
use of oral inosine pranobex and the introduction of diphencyprone as a
contact sensitizer. Good results have been claimed for these treatments
even in severe forms of the disease. We performed a study to investigate
the efficacy of a combination of these treatments in the most severe form
of alopecia areata. Thirty-three patients suffering from alopecia totalis
were enrolled. Subjects were divided into three groups matched for age
and sex. One group received treatment with inosine pranobex (50 mg/kg/day)
for 6 months. The second was sensitized to diphencyprone and treated for
6 months by maintenance of contact allergic dermatitis on the scalp. The
third received both treatments. There was no evidence of response to inosine
pranobex in any of the 22 subjects who received this treatment. Only two
of 22 patients responded to diphencyprone. Patients with long-standing
alopecia totalis contemplating diphencyprone therapy should be advised
that the chances of success are only around 10%. Inosine pranobex does
not appear to improve the response rate.
Hull-SM; Pepall-L; Cunliffe-WJ Alopecia areata in children: response
to treatment with diphencyprone. Br-J-Dermatol. 1991 Aug; 125(2): 164-8.
Twelve children with extensive alopecia areata or alopecia totalis were
treated with the contact allergen diphencyprone. The duration of treatment
ranged from 5 months to 1 year. Eight of the 12 (67%) regrew scalp hair
and in four (33%) there was a complete regrowth. Six months after treatment
was discontinued three of the four children with complete regrowth maintained
their hair, one had lost all the regrowth and a further child with patchy
regrowth at the end of treatment subsequently regrew hair completely while
off therapy.
Van-der-Steen-PH; van-Baar-HM; Perret-CM; Happle-RTreatment of alopecia areata with diphenylcyclopropenone. J-Am-Acad-Dermatol. 1991 Feb; 24(2 Pt 1): 253-7
One hundred thirty-nine patients with severe alopecia areata (the majority with the subtotal, total, or universalis type) were treated with topical immunotherapy (diphenylcyclopropenone). Patients were initially treated unilaterally; the other side of the scalp served as a control. In 50.4% of the patients the response was either excellent (total regrowth) or satisfactory (subtotal regrowth with only a few remaining bald patches). The most frequent side effects were eczematous reactions with blistering, spreading of the induced contact eczema, and sleep disturbances.
van-der-Steen-PH; van-Baar-HM; Happle-R; Boezeman-JB; Perret-CM Prognostic factors in the treatment of alopecia areata with diphenylcyclopropenone. J-Am-Acad-Dermatol. 1991 Feb; 24(2 Pt 1): 227-30
One hundred thirty-nine patients with alopecia areata were treated with diphenylcyclopropenone. Before treatment, 85 patients had subtotal or total hair loss (greater than 90% bald area) and in the remaining patients scalp involvement was between 40% and 90%. The following three factors were found to be of prognostic significance: type of alopecia areata as documented before treatment, duration of the disease before therapy, and presence of nail changes. Other factors such as age at onset, sex, presence of atopic features, the extent of variation in the range of diphenylcyclopropenone concentrations during treatment, and sleep disturbances caused by pruritus did not influence the prognosis significantly.
Naldi-L; Parazzini-F; Cainelli-T Role of topical immunotherapy in the treatment of alopecia areata. Quality analysis of articles published between January 1977 and January 1988 about three treatments. Reading Group.. J-Am-Acad-Dermatol. 1990 Apr; 22(4): 654-6.
We conducted a survey of clinical trials to assess the scientific evidences presented for the practical use of dinitrochlorobenzene, squaric acid dibutylester, and diphencyprone in the treatment of alopecia areata. Twenty-six papers published between January 1977 and January 1988, in English, French, and Italian were selected. We used a standardized protocol of evaluation, which focused principally on the reporting of methods. Twelve papers were uncontrolled studies. Of the controlled studies, 11 had a self-controlled design, two studies involved the use of parallel concurrent controls, and seven were randomized trials. With regard to criteria for entry, follow-up schedules, and criteria for evaluation of response to treatment, the studies were scored generally poorly; therapeutic regimen, patients' characteristics, withdrawals, and description of side effects in these studies were rated more highly. In light of our results, further and better-designed studies are needed for acceptance of dinitrochlorobenzene, squaric acid dibutylester, and diphencyprone in current therapy.
Hull-SM; Norris-JF Diphencyprone in the treatment of long-standing alopecia areata.
Br-J-Dermatol. 1988 Sep; 119(3): 367-74 Thirty-six patients with alopecia areata of 1-54 years duration entered a study of treatment with the contact allergen diphencyprone for 8 months. Following sensitization the diphencyprone was applied to one half of the scalp at weekly intervals, the other half acting as a control. Once hair growth was established on one side, the other side was treated. Seven patients did not continue treatment and one patient showed spontaneous regrowth. Of the remaining 28 patients who persisted with treatments, fourteen (50%) regrew hair on the treated side; eight (29%) had a cosmetically acceptable result with the regrowth of terminal hair over the whole scalp. No statistically significant differences were found in age or duration of alopecia between those who regrew and those who did not. We have found diphencyprone to be an effective stimulator of hair growth in patients with severe and long-standing alopecia areata.
VaN Baar HMJ. Dapsone vs topical immunotherpay in alopecia areata. BJD 1995, 133:270-4.
The percentaje of patients showing regrowth of hair during treatment with dapsone was comparable with the ocurrence of spontaneous regrowth of hair reported in the literature.
Hoffman R, Happle R. Does interleukin -1 induce hair loss?. Dermatology 1995, 191:273-5
abn increased expression of gama interferon, IL-2 and IL1 Beta was found before treatment with the contact sensitizer, whereas IL-10 was the most abundant cytokine present within the scalp after treatment . Of importance was the observation that IL-1Beta was reduced after successful treatment. (JID 1994 103:530-3.)
McElwee KJ, Roston DH Irachy R, Oliver RF. Topical FK506: a potent immunotherapy for alopecia areata?. BJD 1997 137:491-7.
We elected to examine the efficacy of the topical applied immunosuppresive agent FK506 (prograf)in the rtreatment of alopecia areata using the dundee experimental bald rat model. Our findings suggest that FK506 may have considerable potential as a topical treatment for AA.
La dosis menor probada útil en los ratones fue de 0,05 ml de solución de FK506 al 0,1% apliandose 2 dias a la semana (25 microgramos de FK506/cm2)
Mark Lebwohl New treatments for alopecia areata Lancet 1997
For most patients with limited alopecia areata traditional treatments such as topical and intralesional
steroids, minoxidil, and anthralin remain the mainstays
of therapy. Alopecia totalis and universalis are much more refractory to
treatment; the topical preparations offer limited benefit and intralesional
steroid injections only occasionally result in minor regrowth in local
areas. The lack of effective alternatives when the psychologically debilitating
disease of alopecia areata does not respond to established treatments has
led to attempts to add new agents to the dermatologist's range.
Efficacy of PUVA in alopecia areata


PUVA (psoralen plus ultraviolet A) has been used with some success (figure) in patients refractory to other treatments, but there are doubts about whether PUVA is effective in alopecia areata. For example, one of my patients had complete regrowth of hair after 6 months of PUVA--and the local efficacy of PUVA was demonstrated by the regrowth of eyebrow hairs but not of eyelashes, which were covered during UVA exposures. However, once the frequency of treatments was reduced the alopecia returned, indicating that chronic PUVA therapy would be required to maintain remission. The patient therefore had to balance the side-effects of long-term PUVA, including photoageing and carcinogenesis, against the therapeutic response. A 1995 review of 10 years' experience treating 70 alopecia areata patients with PUVA confirmed how difficult it is to maintain the regrowth of hair.1 Up to 50% of patients with alopecia totalis or universalis experienced remission with PUVA, but rapid hair loss occurred in most when PUVA was discontinued; fewer than 15% of patients had a lasting remission.
Topical immunotherapy is another candidate. Dinitrochlorobenzene, squaric acid dibutylester, and diphencyprone (DPCP) have all been used for contact sensitisation. The sensitised patient is then repeatedly exposed to the sensitising chemical, and this regimen has resulted in hair growth in 30-40% of patients in some series. DPCP is not mutagenic in the Ames test and not teratogenic in laboratory animals.2 t also seems to be a superior sensitiser to squaric acid dibutylester. In a recent study of 26 children with alopecia areata, 25 were sensitised with DPCP and then exposed weekly for 3-12 months. Eight children (32%) showed cosmetically acceptable hair regrowth and the regrowth persisted in most patients for at least 6 months.3 Other immunotherapies are being considered. Tumour necrosis factor (TNF-) has been found in inflamed follicles of patients with alopecia areata, so treatment with antibodies to TNF- has been proposed. If a specific target antigen can be identified in this autoimmune disease, it may be reasonable to attempt to induce tolerance by repeated exposure to oral or injected antigens. Therapies directed against the T-cell receptor may be conceivable in the future as well.
S. ahin, B. Yalçn, A. Karaduman PUVA Treatment for Alopecia areata Dermatology 197:3:1998, 245-247.
Twenty-four patients (15 female and 9 male) suffering from
extensive AA for more than 1 year were included in a retrospective
study. PUVA was administered to the whole body 3 times a week.
Excellent hair regrowth was achieved in 37.5% of thepatients.
Uva was administered 3 times a week to the entrire body for 2 h after
intestion of 0.6 mg/kg. 0-MOP. A waldmann 8001 cabinet was used for total
body irradiation. Te initia UVA dose was 0,5 J/cm2. This dose was increased
by 0,5 J/cm2 in each cours to a maximum of 8 J/cm2. Patients were given
a media of 32 courses of therapy. Initial detection of hair regrowth was
observed after 13 courses of therapy.
The median follow-up time after cessation of PUVA treatment was 45
months. Four of the 9 patients who had excellent response relapsed at 3,8,12
and 24 months.
Retrospective study of 68 patients with severe AA (40%scalp
hair loss), treated for at least 5 months. The overall response rate was
70,6%, complete remission was obtained in 30,9% and partial remission in
39%. Among the investigated prognostic factors for the outcome of DCP therapy,
only the extent of AA at the time of initiation of treatment was found
to be of significance.
Oral corticosteroids may be effective in the treatment
of severe alopecia areata(AA), but the side effects of prolonged therapy
limit their use. The benefit of a single intravenous pulse of methylprednisolone
has not been evaluated in patients with ongoing hair loss of less than
12
months' duration. OBJECTIVE: Our purpose was to determine
the effectiveness of an intravenous
pulse of methylprednisolone at 1, 3, 6, and 12 months
in patients with active severe AA of less than
12 months' duration. METHODS: Forty-five patients
were included in this open study. All had rapid and extensive hair loss
for less than 1 year (first occurrence or relapse), with the bald area
exceeding 30% of the scalp. There were 20 multifocal, 10 ophiasic, 9
universalis, and 6 totalis cases. Intravenous methylprednisolone,
250 mg, was administered twice a day on 3 successive days. Follow-up
for at least 12 months (up to 29 months) was performed. The percentage
of pretreatment bald area covered by hair regrowth at 1,3,6, and 12 months
was measured. RESULTS: No majorside effects were observed. Patients with
multifocal AA (n = 20) showed the best response rate, with 9, 12, 13, and
12 showing 100% or 50% to 100% regrowth at 1, 3, 6, and 12 months, respectively.
Relapse occurred at 3 months in 1 patient, at 6 months in 2, and at 12
months in 4. A second pulse was tried in 2 patients with relapse with 100%
regrowth that was stable at 12 and 28 months. In patients with ophiasic
AA (n = 10), no total regrowth was observed; 6 had no response, 4 showed
20% to 70% regrowth at 1 month with relapse at 3 and 6 months. A second
series of pulses was given to the 4 initial responders 3 to 13 months after
the first series; the response rate to this second treatment was better
than the first. In patients with universalis and totalis AA (n = 15), no
total regrowth was observed initially; 8 patients had no response, and
3 showed 50% to 90% regrowth at I month, with subsequent improvement at
3 and 6 months. In 4 patients who did not show an initial response, a significant
(90% to 100%) delayed regrowth was observed between 9 and 16 months after
the pulse therapy. CONCLUSION: A single series of intravenous pulse of
methylprednisolone appears to be well tolerated and effective in patients
with rapidly progressing extensive multifocal AA, but not those with ophiasic
and universalis AA.
Rokhsar CK. Shupack JL. Vafai JV Washenick K. Efficacy of topical
sensitizers in the treatment of alopecia areata. J Am Acad Dermatol 1998
39:751.
| DNCB | Se ha abandonado su uso por sus propiedades mutagénicas en la salmonela y en raton y por su absorción de más del 65 % a traves de la piel del ratón |
| SADBE | Sensibilizante potente. No está presente en el medio natural. Es caro 5 gr:$149. No es estable en acetona como la DPCP. No es mutagénico |
| DPCP (difenciprona) | No mutagénico, sin embargo el metabolito intermedio activado fotoquímicamente puede ser mutagénico. Es muy sensible a la luz y debe ser almacenado en botellas ambar |
| Mecanismo de acción | El mecanismo de acción
es desconocido. Probablementea por medio accion inmunomoduladora. La piel
tratada con sensibilizantes tópicos muestra una reducción
en el cociente CD4/CD8 peribulbar y una disminución de linfocitos
CD6 y de células de Langerhans.
Inicialmente se penso que los sensibilizantes tópicos. Posteriormente se está imponiendo la teroria de la competencia antigénica en la que la inmunoterapia ejerce una efecto inespecifico de inhibición de la respuesta inmune frente a los folículos. No se conoce si estos sensibilizantes ejercen su acción local o sistémicamente |
| Pacientes sin respuesta | 1-2% de pacientes no pueden ser sensibilizados |
| Tolerancia | La sensibilización puede perderse desarrollandose la tolerancia, que incluso puede estar limitada a una sola zona cutánea. La tolerancia se define como el contínuo aumento de la concentración de DPCP hasta alcanzar una concentración del 2% sin la producción de una dermatitis adecuada. La incidencia de tolerancia es del 10,8%. La administración de cimetidina (1 gr/día/mes o 1,2 gr/día unos días) puede reestablecer la dermatitis de contacto y el crecimiento del cabello en pacientes tratados con DNCB, pero no se ha demostrado con DPCP. |
| Respuesta a la DPCP | Se han descrito 17 estudios con
el uso de la DPCP con respuestas entre el 9 y el 85% con una media del
58%. La frecuencia de recidivas esta poco establecida en un seguimiento
de 19 pacientes tratados satisfatoriamente, a los 6 meses el 10% tenían
una pérdida completa del cabello, 43% tenían alopecia en
parches y el 37% mantenían el cabello.
El papel de la inmunoterapia en las alopecias de menos del 40% no está establecido. En un estudio de 119 pacientes con AA de menos del 40% no se encontraron diferencias entre inmunoterapia, minoxidil y placebo. |
| Factores pronóstico | Los factores que implican un factor pronostico negativo más importantes son la duración y la extensión de la enfermedad, seguidos de la edad de inicio y la existencia de antecedentes familiares de AA. Algun estudio implica la afectación ungueal como factor negativo. El desarrollo de dermatitis precoz es un factor positivo. |
| Efectos secundarios | Linfadenopatía cervical dolorosa. Dermaititis localizada o generalizada, reacciones urticriales, coloración amarillenta del c abello, depigmentación cutánea, fiebre y artrálgias. Edema del cuero cabelludo |
|
|
|
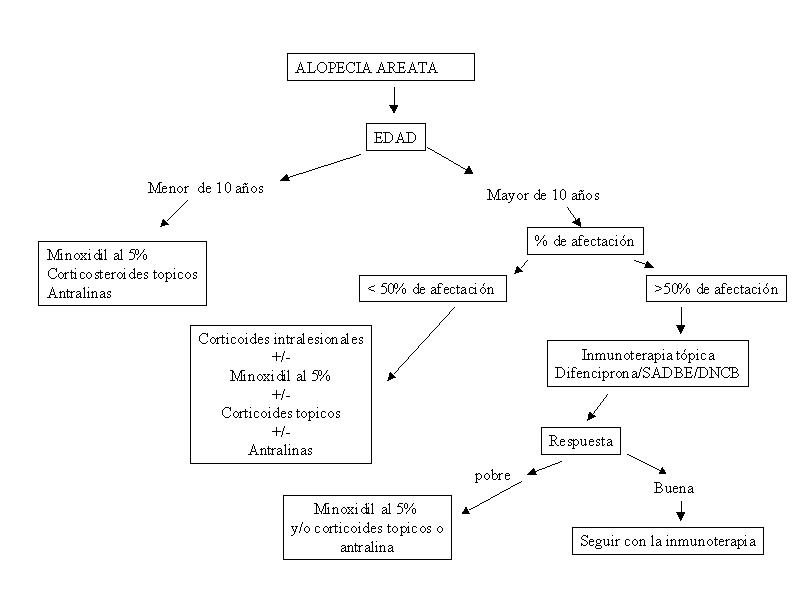
| Patients < 10 years of age | 5% topical minoxidil solution,
topical glucocorticoid, or both
anthralin (short contact (anthralin is lest on scalp for 20 to 60 minutes) |
| Patients > 10 years of age | |
|
<50% scalp afected
|
Intralesional corticosteoid, 5%
topical minoxidil solution, or both, with or without topical glucocorticoid
Anthralin (short contact) |
|
>50% scalp afected
|
5% topical minoxidil solution,
with or without topical glucocorticoid
topical immunotherapy Anthralin (short contact) Oral glucocorticoid Scalp prothesis |
|
Eyebrows and beard affected
|
Intralesional glucocorticoid, 5% topica minoxidil solution, or both |
| The national alopecia areata foundation, 710 C st., Ste 11, Sna Rafael CA 94901-3853 (telephone number, 415-456-4644; Web site http://www.alopeciaareata.com),provides informational brocures, newsletter, resea4ch updates, sources of scalp prostheses, videotapes for schoolchildren and locations of support groups and holds an annual conference to help patients cope with the condition | |
|
|
|
| Intralesional Glucocorticoid | |
| All sites | The prefered compoun is triamcinolone acetonide (10 mg/ml,(concentrations of 2,5-8 mg per ml may also be used)) administered with a 3 ml syringe with a 30-gauge, ½ in.-long needle. Inject 0.1 ml or less into the mid dermis at multiple sites 1 cm apart;do not raise wheal or inject into subcutaneous tissue. Repeat every 4 to 6 weeks; if atrophy of the skin occurs, do not reinject affecte site until atrophy resolves. Optional topical anesthesia may be used: apply a misture of 2,5% lidoicaine and 2.5% prilocina (emla cream) in a thick layer to inctac skin and vover with occlusive dressing for one hour before injections are given; remove cream immediately before injections |
| Scalp | The maximal dose is 20 mg per visti. When more than 50% of scalp is affected, inject onmy selected sites |
| Eyebrows | The maximal dose is 1.25 mg per visit injected into the mid-dermis of each brow at 5 or 6 sites (for a total of 2.5 mg to both brows) |
| Beard | The maximal dose is 7.5 mg per visit |
| 5% topical minoxidil solution | |
| Scalp and beard | The maximal dose is 1 ml per application. Apply twice daily to affected sites. Spread solution with fingers. Wash hands afterward. This treatment is nor effective for patients with total (100%) loss of scalp hair |
| Eyebrows | Apply two applications to each eyebrow with a finger twice daily, using a mirror to ensure precise placement. Hold a cotton ball over the eye for protection. Wash hands afterward. |
| Anthralin (short contact) | (anthralin is left on the scalp
for 20 to 60 minutes)
Apply 0.5 to 1% anthralin cream to affectedd scalp one daily; leave on 20 to 30 minutes daily for 2 weeks, then 45 minutes daily for 2 wk, up to a maximum of 1 hour daily. Wash hands afterward and avoid getting anthralin in the eyes. Remove from scalp with mineral oil, then was off with soap and water. Do not use on brows or beard. Some patients tolerate overnight application. |
| Topical glucocorticoid | Apply twice daily |
| Topical Immunotherapy | |
| Use diphjencyprone or squaric acid dibutyl ester to induce contact sensitization. For initial sensitization , apply 2% solution of selected contact allergen in acetone to a 4-cm2 area on one side of the scalp. After initial sensitization, apply diluted solution of contact allergen weekly to same half of scap in two coats. The patients washes off the allergen after 48 hours after both the sensitizing application and subsequent weekly applications. Adjunst concentrationof allergen according to the response to previous weeks treatment. Deisred responses include mild itching, eritehema and scaling. Concentrations of allergen that elicit responses range from 0,0001% to2%. After hair growth is established on the treated side (in 3-12monts), the both sidesof the scalp are treated. Apply contact sensitizer with wooden application tipped with generous amounto of cotton (the physician or nurse applying weekly treatment must wear gloves). To minimize side effects, it is recommended that the allergen be applied in a physicians office and not given to the patients for use at home | |
| Oral Glucocorticoids | |
| Active, extensive, or rapidly spreading alopecia areata | |
| For patients wighing >60 kg the recommended treatment is 40 mg of oral prednisone daily for 1 wk; then 35 mg daily for 1 wk; 30 mg daily for 1 wk, 25 mg daily for 1 wk, 20 mg daily for 3 days, 15 mg daily for 3 days, 10 mg daily for 3 days and 5 mg daily for 3 days. Prednisona may be used with 5% topical minoxidil solution twice daily and intralesional triamcinolone acetonide injections given as above, every 4 to 6 weeks. Topical therapy should be continued twice daily, with or without intralesional injections every 4 to 6 eeks, after prednisones is tapered. | |
| Active, less extensive alopecia areata | |
| Twenty mg of oral prednisone should be given daily or every other day; dose should be tapered slowly by increments of 1 mg after the condition is stabel | |
Predictive
Model for Immunotherapy of Alopecia Areata With Diphencyprone
Marni C. Wiseman, MD, FRCPC; Jerry Shapiro, MD, FRCPC; Nina
MacDonald, RN, BScN; Harvey Lui, MD
Results Using a survival analysis model, the cumulative patient response at 32 months was 77.9% (95% confidence interval, 56.8%-98.9%). Variables independently associated with clinically significant regrowth were age at onset of disease and baseline extent of AA. Older age at onset of AA portended a better prognosis. A cosmetically acceptable end point was obtained in 17.4% of patients with alopecia totalis/universalis, 60.3% with 75% to 99% AA, 88.1% with 50% to 74% AA, and 100% with 25% to 49% AA. A lag of 3 months was present between initiation of therapy and development of significant hair regrowth in the first responders. Relapse after achieving significant regrowth developed in 62.6% of patients.
Conclusions Response to diphencyprone treatment in AA is affected by baseline extent of AA and age at disease onset. A prolonged treatment course might be necessary. A predictive model has been developed to assist with patient prognostication and counseling.Arch Dermatol. 2001;137:1063-1068




Treatment of severe alopecia areata (AA) remains difficult. To assess the tolerance and efficacy of methotrexate (MTX) in the treatment of severe long-term AA, we retrospectively evaluated 22 patients with AA totalis or universalis with a mean duration of 11.0 +/- 8.8 years who were treated with MTX either alone (n = 6) or associated with low doses of oral prednisone (n = 16). MTX was given at an initial weekly dosage of 15 mg (n = 3), 20 mg (n = 9), or 25 mg (n = 10). Oral prednisone was given at an initial dosage of 10 mg/d in one patient and 20 mg/d in 15 patients. In all, 14 patients (64%) achieved a total recovery including 3 of 6 patients treated by MTX alone and 11 of 16 who had received the combined treatment. Of the 14 patients who had total hair regrowth, 6 stopped MTX. In all, 3 patients maintained hair regrowth and 3 relapsed. Retreatment of these 3 patients by MTX resulted again in hair regrowth. No severe side effect was observed. Although limited by its uncontrolled character, this study shows that MTX and low doses of oral corticosteroids may be an effective and well-tolerated treatment for severe types of AA