Equilibrium 11
We have an equimolar mixture of two species, A2(g) and B2(g). They react at certain temperature, Ta, according to the following equilibrium reaction: A2(g) + B2(g) ⇄ 2 AB(g)
Considering that Kc= 4 for this reaction at Ta, which of the following diagrams best represents the composition of the mixture once the equilibrium has been reached?
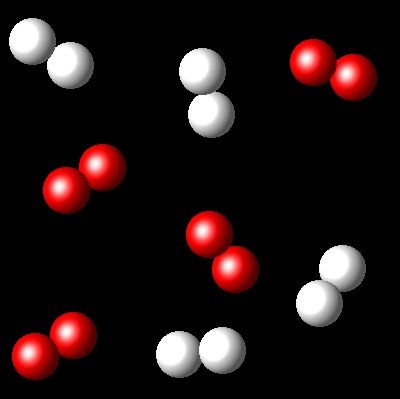 |
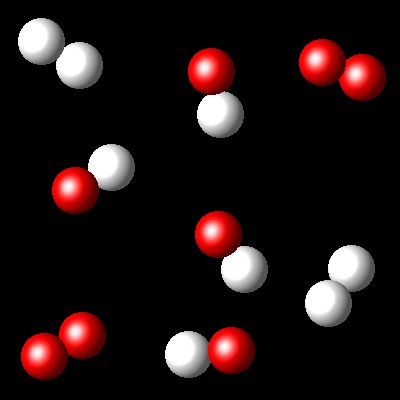 |
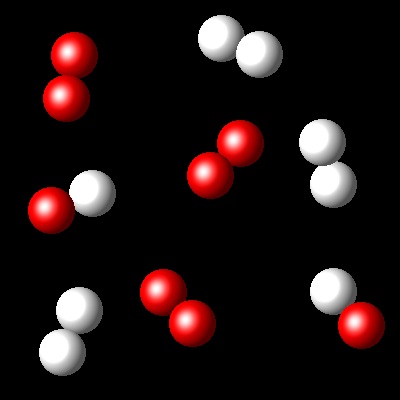 |
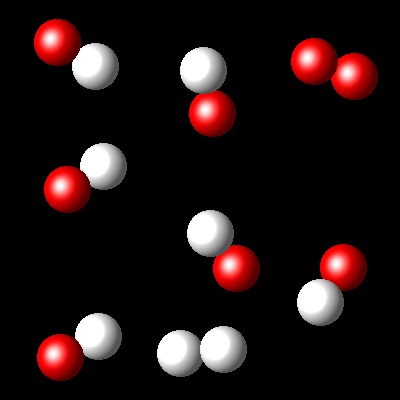 |
Wrong. This diagramme represent zero advance of the reaction.
|
|
Right. This diagram represents Kc=4.
|
|
Wrong. The advance of the reaction that represents
this figure is smaller than what corresponds to a Kc=4.
|
|
Wrong. The advance of the reaction that represents
this figure is bigger than what corresponds to a Kc=4.
|