
information_
Address:
Molecular Science Institute
Coordination Chemistry Group
University of Valencia
Polígono la coma s/n
46980 Paterna
Spain
Some representative results:
Supramolecular coordination chemistry of aromatic polyoxalamide ligands: a metallosupramolecular approach toward functional magnetic materials
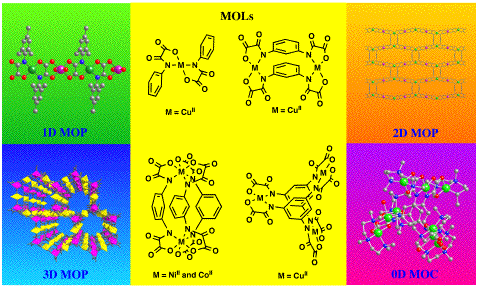
The
impressive potential of the metallosupramolecular approach in designing
new functional magnetic materials constitutes a great scientific
challenge for the chemical research community that requires an
interdisciplinary collaboration. New fundamental concepts and future
applications in nanoscience and nanotechnology will emerge from the
study of magnetism as a supramolecular function in
metallosupramolecular chemistry. Our recent work on the rich
supramolecular coordination chemistry of a novel family of aromatic
polyoxalamide (APOXA) ligands with first-row transition metal ions has
allowed to get on one step further in the rational design of
metallosupramolecular assemblies of increasing structural and magnetic
complexity. Thus, we have taken advantage of the new developments of
metallosupramolecular chemistry and, in particular, the
molecular-programmed self-assembly methods that exploit the
coordination preferences of paramagnetic metal ions and suitable
designed polytopic ligands. The resulting self-assembled di- and
trinuclear metallacyclic complexes with APOXA ligands, either
metallacyclophanes or metallacryptands, are indeed ideal model systems
for the study of the electron exchange mechanism between paramagnetic
metal centers through extended π-conjugated aromatic bridges. So, the
influence of different factors such as the topology and conformation of
the bridging ligand or the electronic configuration and magnetic
anisotropy of the metal ion have been investigated in a systematic way.
These oligonuclear metallacyclic complexes can be important in the
development of a new class of molecular magnetic devices, such as
molecular magnetic wires (MMWs) and switches (MMSs), which are major
goals in the field of molecular electronics and spintronics. On the
other hand, because of their metal binding capacity through the outer
carbonyl-oxygen atoms of the oxamato groups, they can further be used
as ligands, referred to as metal-organic ligands (MOLs), toward either
coordinatively unsaturated metal complexes or fully solvated metal
ions. This well-known “complex-as-ligand” approach affords a wide
variety of high-nuclearity metal-organic clusters (MOCs) and
high-dimensionality metal-organic polymers (MOPs). The judicious choice
of the oligonuclear MOL, reaching from mono- to di- and trinuclear
species, has allowed to control the overall structure and magnetic
properties of the final oxamato-bridged multidimensional (nD, n = 0–3)
MOCs and MOPs. The intercrossing between short- (nanoscopic) and
long-range (macroscopic) magnetic behaviors has been investigated in
this unique family of oxamato-bridged metallosupramolecular magnetic
materials expanding the examples of low-dimensional, single-molecule
(SMMs) and single-chain (SCMs) magnets and high-dimensional,
open-framework magnets (OFMs), which are brand-new targets in the field
of molecular magnetism and materials science (for more details, see the
review paper by M.-C. Dul et al. in Coord. Chem. Rev. 2010, 54,
2281-2296).