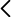- Universitat de València
- Minguez Espallargas, Guillermo
- PDI-Titular d'Universitat
- Coordinador/a de Programa de Doctorat
- Director/a Titulacio Master Oficial
- Romero Pascual, Jorge
- PIT-Tecnic Sup Suport Projecte Investigacio
- Coronado Miralles, Eugenio
- PDI-Catedratic/a d'Universitat
- Director/a d' Institut Universitari
- Javier López Cabrelles
Fuel Cells (FC) are devices that directly convert the energy of a chemical reaction into electricity. Due to the numerous advantages, they show application in the energy sector (high fuel efficiency, clean technology, capable of producing energy for as long as the fuel and oxidant feed the electrodes, etc.). In recent years, there has been a considerable increase in the number of research for its development. There are different types of FC that can be classified according to the temperature or type of electrolyte used.
Among the FC, the Proton Exchange Membranes Fuel Cell (PEMFC) stand out for allowing a low working temperature and a quick start, which requires the use of electrocatalysts to accelerate the cathodic and anodic reactions. However, the catalysts that have been used to date do not show the electrocatalytic activity required to achieve the desired performance. Additionally, they are made of very expensive materials. Therefore, there is a great demand in the development of new electrocatalysts for those applications in the market. Despite the multiple efforts made to advance in the development of these materials, the desired results have not been achieved.
Researchers at the Universitat de València have developed a method to obtain a new catalyst for the electrolysis used by the PEMFC, replacing the current platinum materials, which are much more expensive and scarce. During the process of obtaining the developed nanocomposite, lower cost materials are used. The process was carried out at relatively low temperatures and no solvents are required. The process takes place in short times. In addition, the material obtained is very stable and presents structural characteristics that allow a higher electrocatalytic activity than those reported so far with similar compounds.
Consequently, this new nanocomposite solves the existing problems until now, with the formation of a material with a great electrocatalytic activity. Furthermore, the reduction of production costs responds to the demands of the market.
The invention applies to the energy sector market, mainly to companies that can use the nanocomposite in PEMFC that can be integrated into cars, portable equipment, etc.
The developed nanocomposite material has the following advantages:
- Improvement of electrocatalytic activity. The structural characteristics of the nanocomposite causes an electrocatalytic activity much higher than those reported up to now.
- A considerable reduction in costs, due to the convergence several factors:
- Obtain the precursor with economical materials and in a solvent-free process.
- Obtain the nanocomposite in a relative short time and at relatively low temperature.
- Calcination of the nanocomposite is done using nitrogen, which is much cheaper and safer than other gases used in other processes, such as a mixture of argon and hydrogen.
- Patent granted
Blasco Ibáñez Campus
C/ Amadeu de Savoia, 4
46010 València (València)